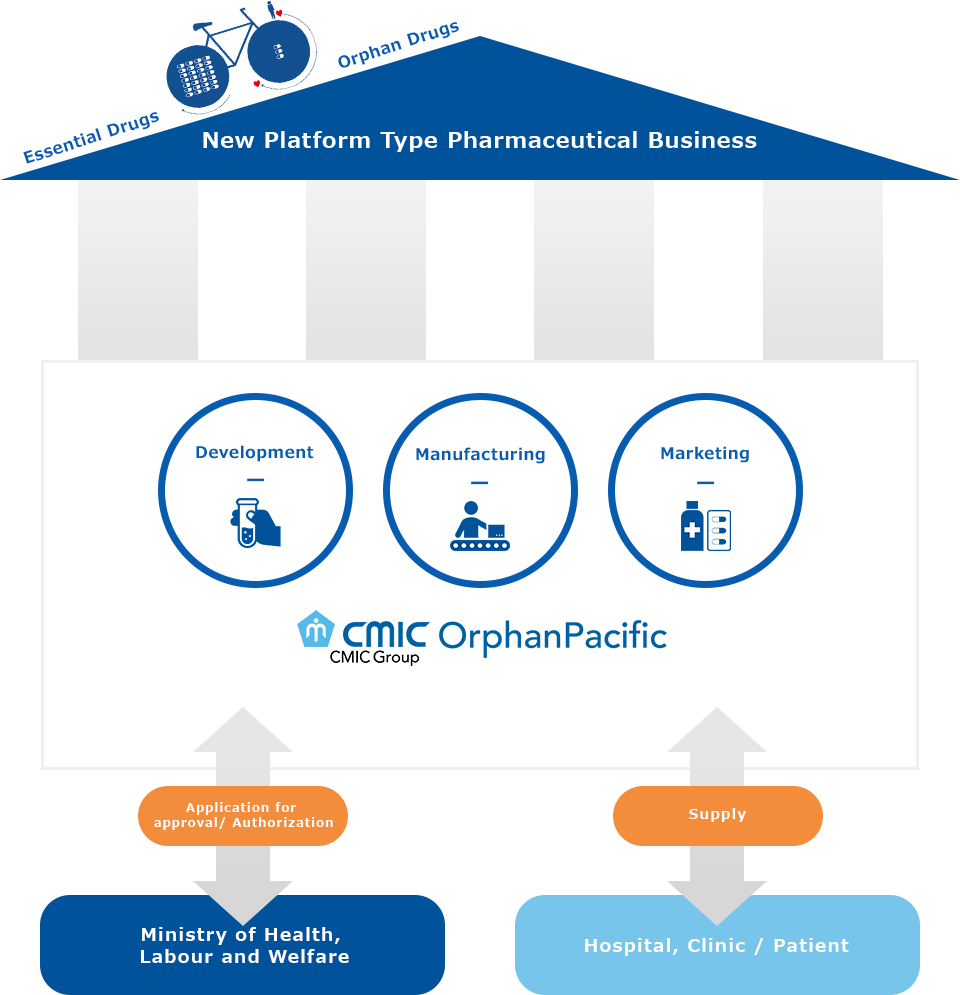
Innovative Pharma Model (IPM)
We support all areas for companies who wish to maximize business in Japan
The various situations where OrphanPacific can prove the right solution
When a company operating in Japan has in mind to transfer the marketing authorization of a prescription drug.
Situation1:
Pharmaceutical companies often face the challenge of prioritization to improve the focus of their organization. Due to strategic considerations, it happens that pharmaceutical companies wish to divest products by transferring their right to other pharmaceutical companies. How could OrphanPacific prove a solution?
Answer 1:
In the few years since its creation, OrphanPacific has accumulated a wealth of experience in regulatory, supply chain, quality management and commercial strategy to run effective product transfer in the field of orphan drugs (e.g. SOMAZON) or long-listed products (e.g. DANTRIUM, ASTOMIN) and the transfers operated so far never caused confusion or critical situations in the medical field, thanks to an efficient coordination with wholesalers, a motivated sales force and a high-performance call center.
Situation 2:
When a pharmaceutical company is primarily focusing on medical affairs and development but would like to outsource post-marketing surveillance and promotion in the field of orphan drugs, why is OrphanPacific a good choice?
Answer 2:
From its creation, OrphanPacific has been involved in the efficient management of EPPV (early phase pharmaco-vigilance) and PMS (post-marketing surveillance) thanks to products like NORMOSANG or BUPHENYL. Developing strong links with specialists by the provision of tailored medical and scientific information to support the optimal usage of our products, we have gained a good reputation in the field of rare diseases.
When a foreign company plans to find a partner in Japan.
Situation:
As an established R&D organization overseas, a foreign company contemplates launching an orphan drug in Japan but have no affiliate of legal presence in Japan to proceed through the regulatory process of submission, approval, pricing negotiation and terms of supply in the fields via wholesalers. How and why OrphanPacific could be the right solution for the company?
Answer:
OrphanPacific has built a robust regulatory affairs management system with the support of its mother company CMIC Holdings to provide continuous and specific advisory support to foreign companies willing to introduce new products to the Japanese market. If the situation requires, OrphanPacific offers the possibility to hold the license on behalf of the originating company while operating on its behalf in terms of promotion and medical development. OrphanPacific has an experienced team in drug safety data management that is used to cooperate with firms in Europe and in the US to guarantee the timely and accurate reporting of adverse drug reactions.
When a bio-venture or academic research organization plans to develop a new drug.
Situation:
A bio-venture or academic research organization is primarily focusing on basic research but would like to pursue the feasibility of the new drug through formulation design, translational research and clinical development. How OrphanPacific can assist them in this case?
Answer:
Internally, OrphanPacific organization includes development and regulatory experts so it can guarantee the planning and the coordination of operations. Externally, OrphanPacific is the affiliate of CMIC Holdings, a group composed of expert organizations in various fields covering drug formulation design, clinical development planning and study execution, consultancy in registration and pricing strategy, and OrphanPacific can work closely with all those parties to optimize the time-to-market as well as the quality of the registration file. Beyond approval, OrphanPacific main shareholders also include MEDIPAL Holdings, the leading drug wholesale organization in Japan, and it is possible to deliver the products across the whole market in compliance with Japanese Good Supplying Practice (JGSP).
When a new pharmaceutical company is planned to be established.
Situation:
You have a plan to create a pharmaceutical company in Japan, how could OrphanPacific be of help in this project?
Answer.
OrphanPacific can provide support to your plan of creation of a licensed pharmaceutical business while effectively running the commercial, regulatory, and medical business, with a framework of progressive disengagement as your new business gets more resources and staffing. OrphanPacific experts’ team will be able to extend support and training to newly formed organization to ensure a smooth transfer of competency.