Market Access
Drug Pricing and Reimbursement
Japan has a unique pricing system for ethical drugs. Understanding this system is crucial when strategizing for a successful launch in Japan.
Opportunities
-
In Japan, it is a principle to include drugs in the NHI (National Health Insurance) drug price list within 60 days (up to 90 days) after approval, and it is mandatory to launch the product within 3 months of price listing. This ensures a prompt process from approval to reimbursement, with clear timeframes.
-
Japanese NHI system allows drugs listed in the NHI drug price list to be accessed with patient co-payments ranging from 10% to 30%, facilitating easy access to ethical drugs. For designated intractable diseases or chronic pediatric specific diseases, financial assistance is available to reduce patients' monthly out-of-pocket costs.
Key Challenges
-
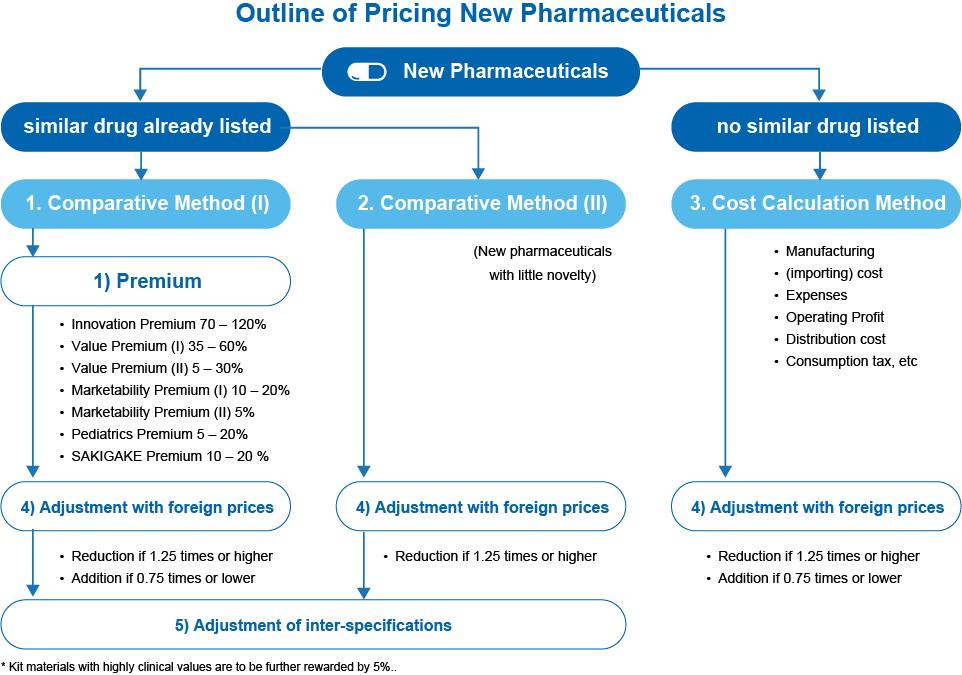
For new drugs (new molecular entities), price listing occurs four times a year (as of September 2024). The price is determined based on the cost calculation method or the similar drug comparison method, with various additional factors applied.
-
Reference prices from the United States, United Kingdom, Germany, and France are considered, making global market plans important.
-
The review results issued at the time of new drug approval, including efficacy, safety, and utility, may influence the pricing, so it is crucial to consider pricing strategies from the development stage.
-
Although a Health Technology Assessment (HTA) system is in place, orphan drugs are generally not subject to HTA (with some exceptions).
Source: PMDA https://www.pmda.go.jp/files/000221888.pdf
If you would like to know more about pricing system in Japan,
Early Access in Japan
In overseas countries, programs like Named Patient Use exist, but Japan's regulatory system is different. Understanding access to ethical drugs for patients with rare diseases who have limited treatment options is important.
-
In Japan, a "mixed practice" of insurance-covered treatment (listed drugs, etc.) and non-insurance-covered treatment (unapproved drugs, etc.) is generally not permitted. If non-insurance-covered treatment is included, the patient bears all medical expenses. Therefore, it is desirable to proceed with development and regulatory approval quickly.
-
However, there are exceptions, and the following means are recognized in Japan as Early Access options:
- Before approval:
-
-
Clinical trials (Company-sponsored trials, Investigator-initiated trials) : Expanded Clinical Trials are designed as a framework for compassionate use.
-
Clinical research (Specified Clinical Research)
-
Evaluation Treatment
-
- After approval:
-
-
Clinical research (Specified Clinical Research)
-
Evaluation Treatment
-







