Regulatory Systems in Japan
Regulatory Approval for New Drugs
Summary of Regulatory Approval in Japan
1.Regulatory Approval for New Drug
-
PMDA (Pharmaceuticals and Medical Devices Agency): Conducts review of application dossiers and conducts compliance inspections
-
MHLW (Ministry of Health Labour and Welfare):Approval authority
-
Expert Discussion:PMDA review team members and external experts to discuss important issues.
-
Drug Committees:An advisory body of the MHLW providing expert advice on drug approval.
2. Process for Regulatory Approval
The approval process is outlined in the figure below. Alongside the review of application documents, GLP/GCP & data integrity assessments, and GMP inspections are conducted by PMDA.
After approval, the "Review Report" is published on the PMDA website.

Source: PMDA https://www.pmda.go.jp/files/000153667.pdf
3. Review Timeline for New Drugs
-
Priority Review
-
Orphan Drugs* : 9 months (median)
*if the drug is applicable for priority review
-
Drugs designated as “Sakigake”: 6 months (with pre-review)
-
-
Standard Review: 12 months
-
Specific-purpose Drugs
4. Approval Timing
Generally, it is eight times a year within three weeks after Drug Committees (from 2025).
However, it is seven times in the year preceding the drug price revision.
NDA Submission and Approval (MAH)
Applicant of NDA Submission and Approval
To obtain regulatory approval for a new drug, a company must have a pharmaceutical business license. Regulatory approval for individual drugs is granted to companies this. There is also a system called “Foreign Exceptional Approval System“* as an exception.
*The system that allows foreign companies to obtain pharmaceutical approval in Japan.
Applicant of New Drug Application
-
General Regulatory Approval: Companies with a pharmaceutical business license
-
Foreign Exceptional Approval: Overseas pharmaceutical companies (However, it is necessary to appoint a domestic company with pharmaceutical business license in Japan.)
Key Challenges
To obtain pharmaceutical business license, it is necessary:
-
To have a “TRIAD” (“San-yaku” in the below chart; General Marketing Compliance Officer, Quality Officer, Safety Officer) in a Japanese corporation (see Figure). However, securing such talent is increasingly difficult.
-
To establish GVP (Good Vigilance Practice) and GQP (Good Quality Practice) systems, and have necessary Standard Operating Procedures (SOPs) to comply with regulations
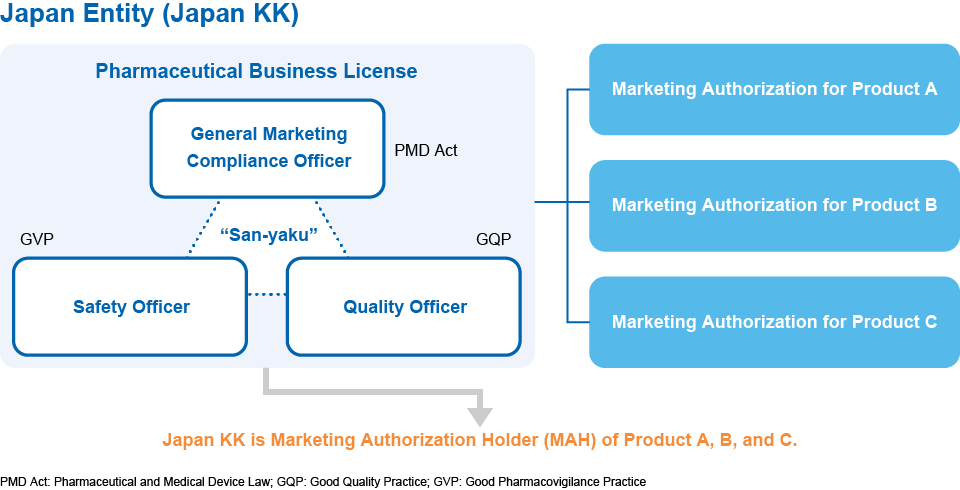
MAH and AMAH
Under Japanese laws and regulations, Marketing Authorization of a drug is granted to the pharmaceutical company with Japanese pharmaceutical business licenses. However, even if a foreign company does not establish a Japanese company with pharmaceutical business license, it can apply for marketing approval using the foreign exceptional approval system by appointing OrphanPacific as Appointed Marketing Authorization Holder (“AMAH”). AMAH is responsible to MHLW and PMDA on behalf of foreign pharma.

Role & Responsibility of MAH and AMAH
| MAH | Foreign Exceptional Approval Holder/AMAH | |
|---|---|---|
| Applicant for NDA | OrphanPacific | Foreign Pharma (AMAH is stated in the applicant form) |
| Approval (Marketing Authorization) |
OrphanPacific | Foreign Pharma (Foreign exceptional approval) |
| Corresponding to MHLW and PMDA |
OrphanPacific | OrphanPacific will be a contact window to MHLW and PMDA while communicating with the client |
| Definitive Responsibilities | OrphanPacific | Foreign Pharma |
| Regulatory Responsibility | OrphanPacific | OrphanPacific |
Clinical Development for NDA
Even for rare diseases with a small number of patients, clinical trial data from Japanese subjects is generally required (as of September 2024). Participation in global clinical trials is also an option, but it is important to note that diagnostic criteria and standard treatments may differ from those overseas, making prior research and planning crucial.
Opportunities
There is a system in place that allows overseas pharmaceutical companies without a Japan entity to conduct clinical trials by asking CRO to be In-Country Clinical Caretaker (ICCC).
Key Challenges
Clinical trial data from Japanese subjects is generally required. Differences in diagnostic criteria and standard treatments may complicate participation in global trials.
If you would like to know more about NDA in Japan,
Orphan Drug Designation (ODD)
In Japan, an Orphan Drug Designation (ODD) system is available.
Opportunities
Receiving ODD offers benefits such as priority review (median 9 months), extension of re-examination period (market exclusive period, up to 10 years), and price premium (marketability premium I: 10-20%).
Key Challenges
Steps to complete before receiving incentives:
-
Conduct ODD consultations and discuss with MHLW to determine eligibility.
-
If eligible, submit application materials, undergo committee review, and complete the designation process.
Since the criterion for designation includes “Possibility of Development,” more designations occur in the late phase compared to overseas. Completion of the Investigational New Drug (IND) application in Japan often required.
Designation Criteria
-
Number of Patients: Less than 50,000 in Japan
-
Medical Needs: If marketing authorization is granted for the pharmaceutical product, it is expected to have particularly excellent utility for its intended use. In principle, it must meet the following criteria 1 and 2, indicating a high medical necessity;
-
Severity of the target disease
-
The usefulness for the target disease, satisfying one of the following criteria 1) to 3);
- No approved drugs and related products available.
- Even though approved drugs and related products are available, these approved drugs etc. alone are not sufficient for treatment or prevention, and other multiple options are clinically required.
- Higher efficacy or safety is expected compared with existing products.
-
-
Possibility of Development: Having the capability and plan to conduct development domestically. Specifically, having a clear overview of the clinical trials planned to be conducted up to the point of submitting the approval application. Additionally, having largely completed the necessary non-clinical trials required to conduct the first clinical trial involving human subjects.
Incentives
-
Subsidy for R&D
-
PMDA priority consultation on R&D*
-
Tax exemption: 20% of R&D cost during subsidy payment period
-
Priority Review* (median 9 months for PMDA review)
-
Reduction of examination fees
-
Extension of re-examination period (up to 10 years)
*Eligible for priority review or consultation
Regulatory Authorities for ODD
-
MHLW:Designation Authority
-
PMDA:Priority Scientific Consultation for clinical trial and CTD
-
NIBIO: Subsidy payment etc.
If you would like to know more about Orphan Drug Designation in Japan,
Other Regulatory Systems
Sakigake Designation System
The SAKIGAKE Designation System aims to designate innovative drugs developed ahead of the world for early commercialization in Japan, with notable efficacy expected during early clinical trials.
Designation Criteria
-
Innovativeness of the treatment
-
Severity of the target disease
-
Extremely high efficacy for the target disease
-
Intention and infrastructure to develop and apply early in Japan, ahead of the world (including simultaneous application)
Benefits
-
Priority consultation
-
Expedited pre-submission review (Rolling review)
-
Priority review (12 months → 6 months)
-
Examination Partner System (concierge service by PMDA)
-
Enhancement of post-marketing safety measures (extension of re-examination period: up to 10 years)
Conditional Early Approval System
For pharmaceuticals treating serious conditions with few effective options, as well as those that are difficult to conduct confirmatory clinical trials due to reasons such as a small number of patients or requiring a long period of time, it is possible to ensure prompt patient access to drugs with high medical needs by approving them with conditions for necessary investigations to confirm efficacy and safety after marketing. A mechanism is introduced to promptly evaluate the results of post-marketing surveillance and other investigations conducted with conditions applied, and to reflect them in safety measures, etc.
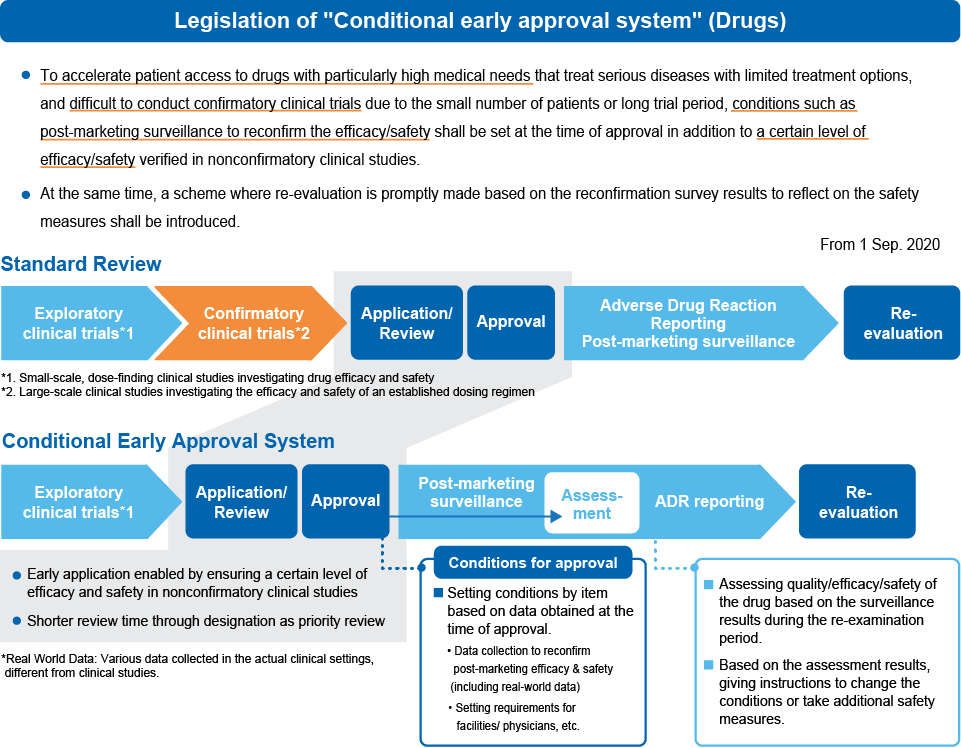
Source: PMDA https://apac-asia.com/images/achievements/pdf/7th/A_Regulations/RA-06_Introduction.pdf







