Post-Approval Commitments (Safety)
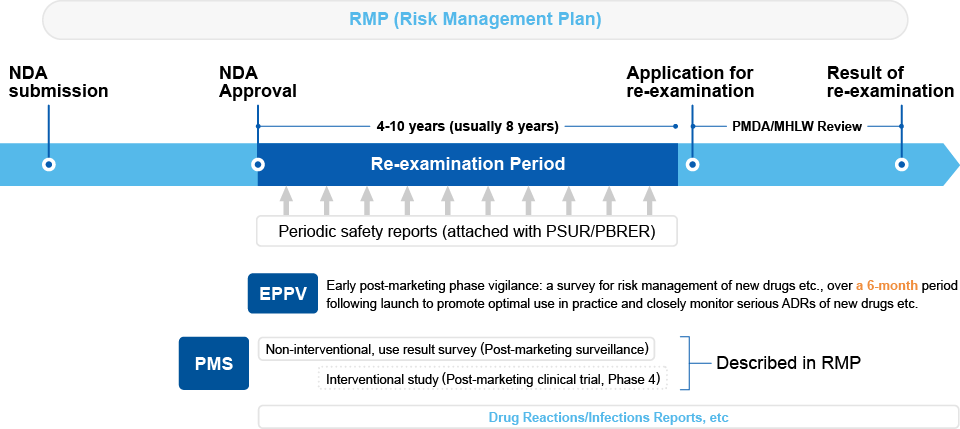
Re-examination System
-
To confirm the effectiveness and safety of new drugs, Japan has established a "re-examination system". During the re-examination period, generic drugs cannot enter the market, resulting in a period of substantial market exclusivity.
-
It is essential to collect post-marketing surveillance (PMS) data within the specified period set at the time of approval and undergo re-examination by the Pharmaceuticals and Medical Devices Agency (PMDA).
-
The re-examination periods vary, such as 8 years for new molecular entities (NMEs, excluding orphan drugs) and 10 years for orphan drug NMEs.
Pharmacovigilance and Post-Marketing Surveillance
As in other countries, safety monitoring activities are conducted based on the Risk Management Plan (RMP).
- There are two unique systems in Japan:
-
-
EPPV (Early Post-Marketing Phase Vigilance):
This is implemented to intensively collect safety information immediately after the product is marketed. For the first two months, Medical Representatives (MRs) visit healthcare institutions every two weeks, followed by monthly visits until six months from the launch to collect safety information. -
PMS (Post-Marketing Surveillance):
For widely used drugs, it is required to collect information on approximately 3,000 cases of product use after market approval. In the case of orphan drugs with a small number of patients, often all cases are investigated, and the investigation period and target number of cases are determined through consultations with the PMDA.
-