Our Capability in the Orphan Drug Business
Since its establishment, OrphanPacific has been diligently working to deliver orphan drugs to rare disease patients in Japan as quickly as possible. We have experience in the asset acquisition, development, regulatory approval, and sales of the following products, including ultra-orphan drugs (for patient populations of 1,000 or fewer).
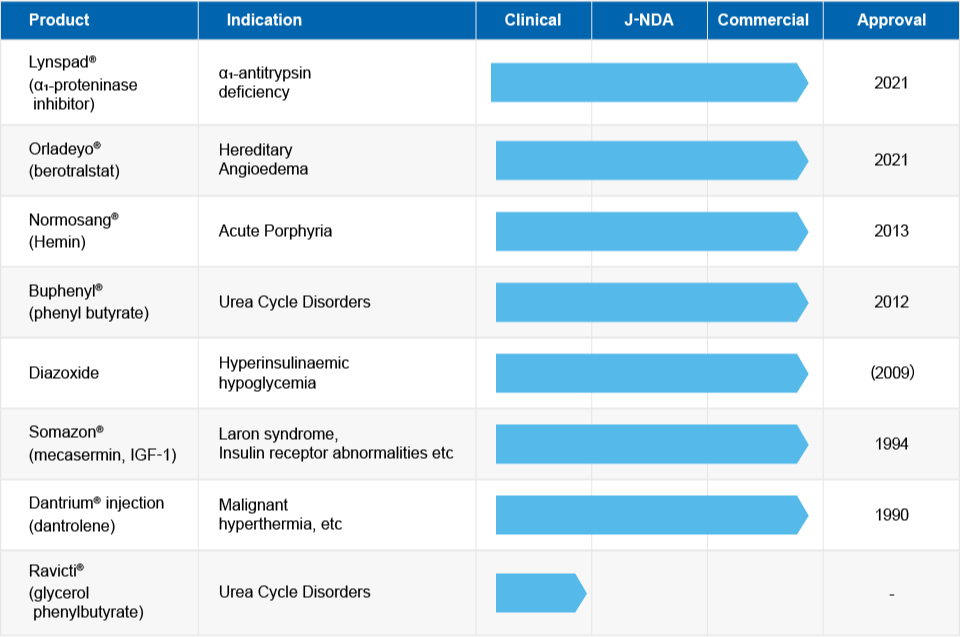
Many orphan drugs approved overseas or in late development stages have not been developed domestically in Japan. OrphanPacific actively pursues in-licensing and partnerships with overseas companies to promote the development and sales of these pharmaceuticals in Japan.
OrphanPacific has extensive experience collaborating with many overseas companies and is well-versed in the differences in regulations and business practices between Japan and other countries, including clinical development, manufacturing, and quality management in the pharmaceutical industry. Leveraging overseas data and know-how, OrphanPacific has the experience and expertise to efficiently develop and market orphan drugs in Japan.







